First Alzheimer’s Disease Patient Enrolled in Clinical Trial at Columbia University
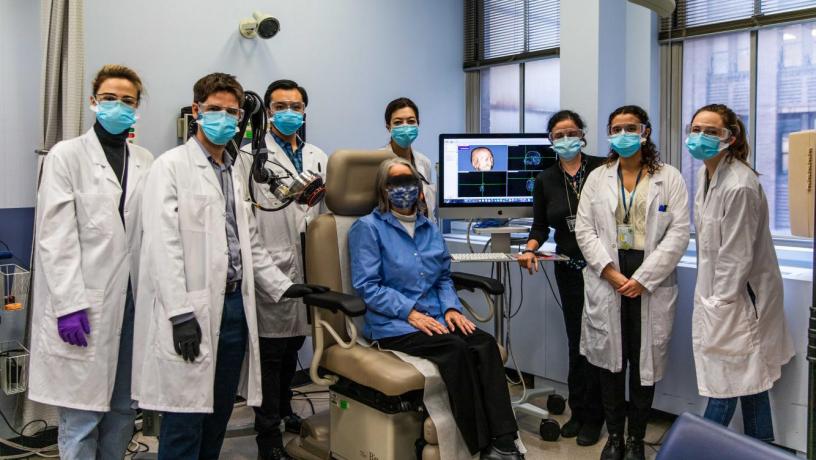
The research team and the subject in the outpatient room where the procedure took place. The ultrasound transducer was attached to a robotic arm and was guided by the neuronavigation system via an infrared camera.

The robotic arm and ultrasound transducer positioned on the subject's scalp.

The research team and the subject in the MRI suite following confirmation of the non-invasive and targeted blood-brain barrier opening.
Elisa Konofagou, PhD, the Robert and Margaret Hariri Professor of Biomedical Engineering and Radiology at Columbia University, and her research team recently treated the first patient in their clinical trial to open the blood-brain barrier (BBB) in patients with Alzheimer’s disease.
The team, based out of Dr. Konofagou’s Ultrasound Elasticity Imaging Laboratory (UEIL), is using a novel, neuronavigation-guided focused ultrasound system that they designed and tested.
The clinical trial will now enroll up to six eligible patients to assess the safety and feasibility of focused ultrasound–induced BBB opening with a single-element transducer under neuronavigational guidance. Using MRI and positron emission tomography, the investigators will also test whether the focused ultrasound procedure decreases the amyloid protein levels in the treated brain areas. Finally, the team plans to evaluate any potential treatment effects on the patients’ cognitive function.
Dr. Konofagou said, “I feel very proud of my incredible team for making a 16-year-old dream come true yesterday…A big thank you to the team for their perseverance and hard work as well as to the several UEIL alumni whose dedication and important discoveries have been the key to this accomplishment…”.
"Over the past 15 years," Konofagou continued, "we have been working on a technique that opens the blood-brain barrier, which is one of the main hurdles in brain drug delivery. We have shown that we can get drugs in safely that can treat Alzheimer’s and Parkinson’s as well as brain tumors in mice. On November 2, 2020, after 3 years of fighting the FDA followed by the pandemic, we were able to translate our technique to the clinic, having started our clinical trial in treating Alzheimer’s patients. The barrier was opened safely in a patient with Alzheimer’s and preliminary findings indicate a potential reduction in amyloid, one of the proteins that accumulates in the brains of Alzheimer’s patients."
Antonios Pouliopoulos, UEIL associate research scientist and one of the team's leaders of the study, added that, "the ultrasound system used in this trial was designed and pre-clinically tested by our team. It is a simple, portable, flexible, cost-effective system and can achieve millimeter-precise opening of the blood-brain barrier in a fully non-invasive and painless manner. It also allows for real-time monitoring of the treatment. The whole procedure can be completed in approximately 30 minutes and the patient is discharged the same day. It is worth mentioning that this is a purely academic effort here at Columbia, without the support of a company. We believe that our approach can revolutionize the way brain diseases are treated and this trial is a pivotal step for the wider adoption of therapeutic ultrasound throughout the world."
The study is co-led by Dr. Konofagou and Lawrence Honig, MD, PhD, a neurologist at the Taub Institute of Alzheimer’s Disease and Aging at Columbia. The Focused Ultrasound Foundation is providing funding for this study.